Two Hydrogen Atoms Form A Hydrogen Molecule When
Two Hydrogen Atoms Form A Hydrogen Molecule When - Web this type of chemical bond is called a covalent bond. How atoms interact with other atoms is largely dependant on the number of electrons on its outermost layer. The single electrons from each of the two hydrogen atoms are shared when the atoms come together to form a. Two i atoms can form an h2. They are formed when certain hydrogens attract atoms in other molecules such as oxygen. The bond is represented either as a pair of “dots” or as a solid line. It’s a diatomic gas as opposed to a monatomic gas (like helium). The simplest case is a pair of water molecules with one hydrogen bond between them, which is called the water dimer and is often used as a model system. Web a water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent. Hydrogen is the lightest element.
The single electrons from each of the two hydrogen atoms are shared when the atoms come together to form a. The simplest case is a pair of water molecules with one hydrogen bond between them, which is called the water dimer and is often used as a model system. Hydrogen is the lightest element. Web hydrogen bonds are not formed when two hydrogens attract: The two hydrogen atoms are attracted to the same pair of electrons in the covalent bond. Two i atoms can form an h2. The fact that water has two hydrogens attached to a single oxygen is determined by the number of bonds that an oxygen atom can form. A covalent bond is a bond in which two atoms share one or more pairs of electrons. A gas can never be an element. It’s a diatomic gas as opposed to a monatomic gas (like helium).
This is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons. A covalent bond is a bond in which two atoms share one or more pairs of electrons. Web this type of chemical bond is called a covalent bond. Web hydrogen bonds are not formed when two hydrogens attract: Web a water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent. Same deal for oxygen (gas = o 2 ). Web hydrogen the molecule (hydrogen gas) has two hydrogen atoms bonded together to form h 2. But if the hydrogen atom no. Web in a discrete water molecule, there are two hydrogen atoms and one oxygen atom. A gas can never be an element.
How to Learn About the Chemistry of the Hydrogen Atom 12 Steps
This is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons. They are formed when certain hydrogens attract atoms in other molecules such as oxygen. Web a hydrogen molecule forms from two hydrogen atoms, each with one electron in a 1 s orbital. When two atoms of hydrogen combine.
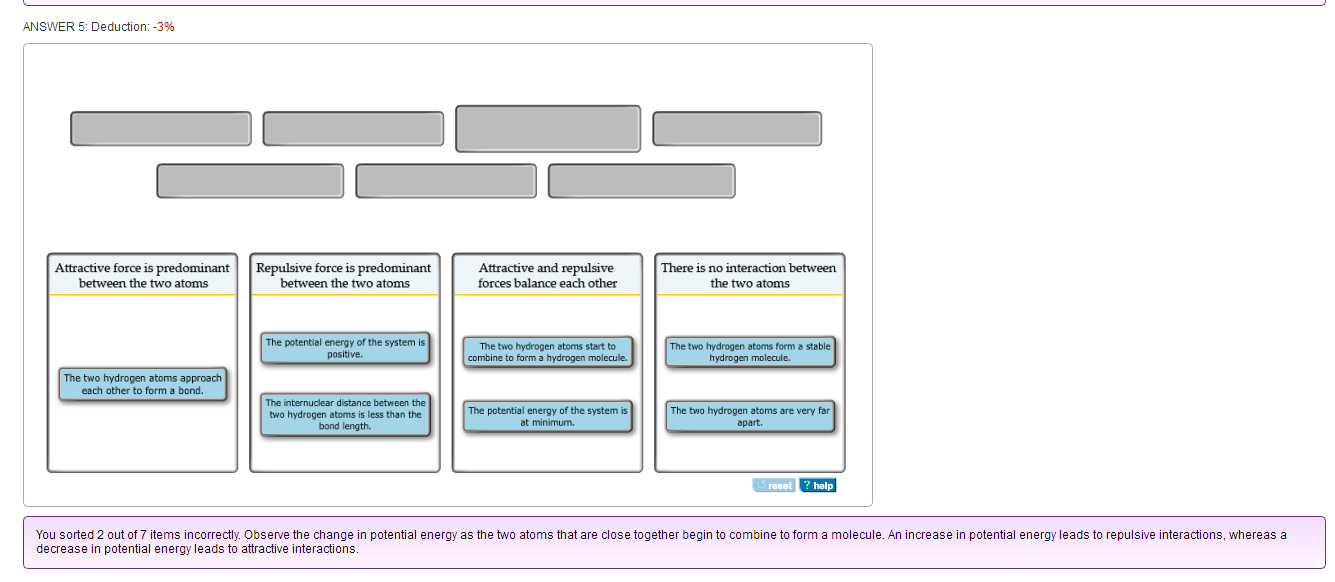
Two hydrogen atoms interact to form a hydrogen molecule. Classify the
Web hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons; A gas can never be an element. Web hydrogen the molecule (hydrogen gas) has two hydrogen atoms bonded together to form h 2. Web when two hydrogen atoms fuse with one oxygen atom to create a molecule of water,.
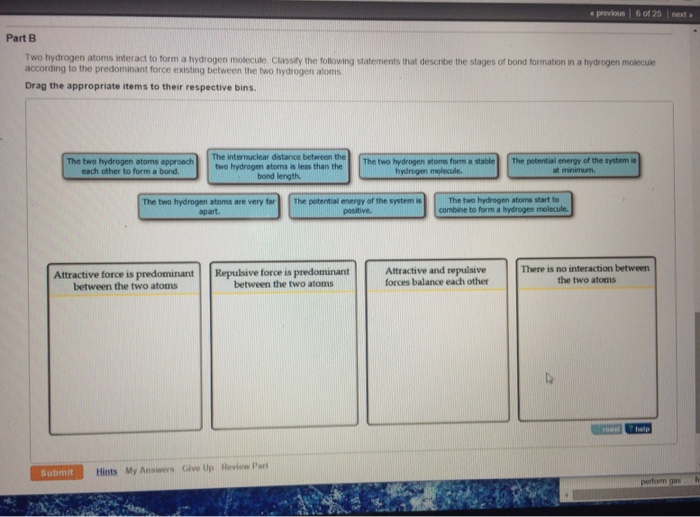
Solved Two hydrogen atoms interact to form a hydrogen
Two i atoms can form an h2. Same deal for oxygen (gas = o 2 ). Web hydrogen bonds are not formed when two hydrogens attract: Web when two hydrogen atoms fuse with one oxygen atom to create a molecule of water, each hydrogen atom donates its single electron to the oxygen atom, resulting in 10 electrons for the oxygen,.
Wasserstoffmolekül H2 / hydrogen molecule H2 Stock Photo Alamy
Web answer (1 of 5): A covalent bond is a bond in which two atoms share one or more pairs of electrons. Web hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons; Web in a discrete water molecule, there are two hydrogen atoms and one oxygen atom. This is.
Chemistry model of molecule hydrogen H2 scientific element. Integrated
Web hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons; Web hydrogen bonds are not formed when two hydrogens attract: This is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons. Web i was wondering (with my limited.
Two hydrogen atoms interact to form a hydrogen molecule. Classify the
Two i atoms can form an h2. That just means each particle of gas is a molecule comprising two atoms rather than single atoms. It’s a diatomic gas as opposed to a monatomic gas (like helium). The two hydrogen atoms are attracted to the same pair of electrons in the covalent bond. Web a water molecule consists of two hydrogen.
Hydrogen Molecule Air Water Life®
But if the hydrogen atom no. An element is a classification of atoms based on the. Such a bond is weaker than an ionic bond or covalent bond but stronger than van der waals forces. Web i was wondering (with my limited classical physics knowledge) why two hydrogen atoms tend naturally to a bonding configuration, i mean, given two hydrogen.
Bonds That Hold Water Molecules Together / Intermolecular Forces
The simplest case is a pair of water molecules with one hydrogen bond between them, which is called the water dimer and is often used as a model system. Two i atoms can form an h2. How atoms interact with other atoms is largely dependant on the number of electrons on its outermost layer. Web in a discrete water molecule,.
Solved Two hydrogen atoms interact to form a hydrogen
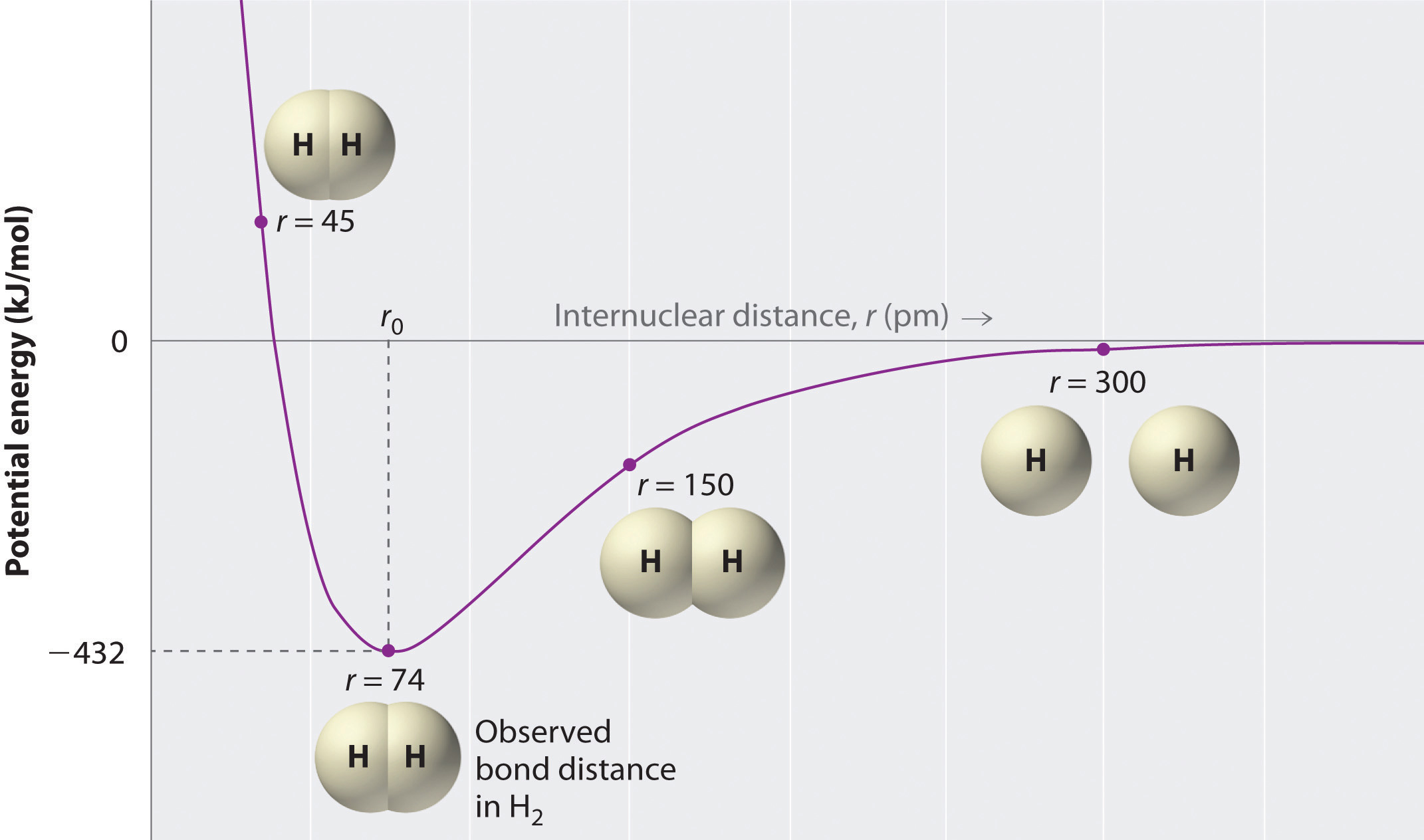
Well the atoms are not exactly neutral. Web hydrogen the molecule (hydrogen gas) has two hydrogen atoms bonded together to form h 2. As you may know, all atoms are comprised of a small nucleus surrounded by 'cloud' of electrons. When two atoms of hydrogen combine to form a molecule of hydrogen gas, the energy of the molecule is lower.
Blog Hydrogen offers huge potential economic opportunity for Alberta
Same deal for oxygen (gas = o 2 ). How atoms interact with other atoms is largely dependant on the number of electrons on its outermost layer. Web i was wondering (with my limited classical physics knowledge) why two hydrogen atoms tend naturally to a bonding configuration, i mean, given two hydrogen atoms with zero relative velocity between eachoter, and.
As You May Know, All Atoms Are Comprised Of A Small Nucleus Surrounded By 'Cloud' Of Electrons.
Web in a discrete water molecule, there are two hydrogen atoms and one oxygen atom. Web i was wondering (with my limited classical physics knowledge) why two hydrogen atoms tend naturally to a bonding configuration, i mean, given two hydrogen atoms with zero relative velocity between eachoter, and given that a monoatomic. The fact that water has two hydrogens attached to a single oxygen is determined by the number of bonds that an oxygen atom can form. How atoms interact with other atoms is largely dependant on the number of electrons on its outermost layer.
Same Deal For Oxygen (Gas = O 2 ).
A gas can never be an element. The single electrons from each of the two hydrogen atoms are shared when the atoms come together to form a. Two i atoms can form an h2. Such a bond is weaker than an ionic bond or covalent bond but stronger than van der waals forces.
Web Answer (1 Of 5):
They are formed when certain hydrogens attract atoms in other molecules such as oxygen. Web when two hydrogen atoms fuse with one oxygen atom to create a molecule of water, each hydrogen atom donates its single electron to the oxygen atom, resulting in 10 electrons for the oxygen, instead of eight. At standard conditions hydrogen is a gas of diatomic molecules having the formula h2. Web hydrogen the molecule (hydrogen gas) has two hydrogen atoms bonded together to form h 2.
That Just Means Each Particle Of Gas Is A Molecule Comprising Two Atoms Rather Than Single Atoms.
Web a hydrogen molecule forms from two hydrogen atoms, each with one electron in a 1 s orbital. Web a water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent. The electrons on this layer are called. This is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons.